surya kiran
Regular Member
- Joined
- Jun 17, 2013
- Messages
- 33
- Likes
- 135
- Limited supply and steep price of the two approved Covid-19 antivirals driving many Chinese to opt for cheaper but illegal imports from India
- Indian generics have not been approved by the Chinese government and selling them is a punishable offence
Chinese residents have turned to the black market for generic Covid-19 drugs, as an unprecedented pandemic outbreak ramps up demand for limited supplies of approved antivirals.
China approved two Covid-19 antivirals this year – Pfizer’s Paxlovid and Azvudine, an HIV drug from Chinese firm Genuine Biotech. But both are only available in certain hospitals.
Their limited supply and steep price have driven many Chinese to opt for cheaper but illegally imported generic drugs from India.
Topics like “anti-Covid Indian generic drugs sold at 1,000 yuan (US$144) per box” have been trending on Chinese social media platform Weibo, with users exchanging messages and tips on ways to get hold of the medicines.Four kinds of generic anti-Covid drugs from India are being sold illegally in the Chinese market – under the brand names Primovir, Paxista, Molnunat and Molnatris.
While Paxlovid is priced at 2,980 yuan per box, a box of Indian-made drugs can be bought for 530 to 1,600 yuan, according to online portal Tencent News. Primovir and Paxista are generic versions of Paxlovid, while Molnunat and Molnatris are generics for Merck’s molnupiravir.
However, the Indian generics have not been approved by the Chinese government and selling them is a punishable offence.
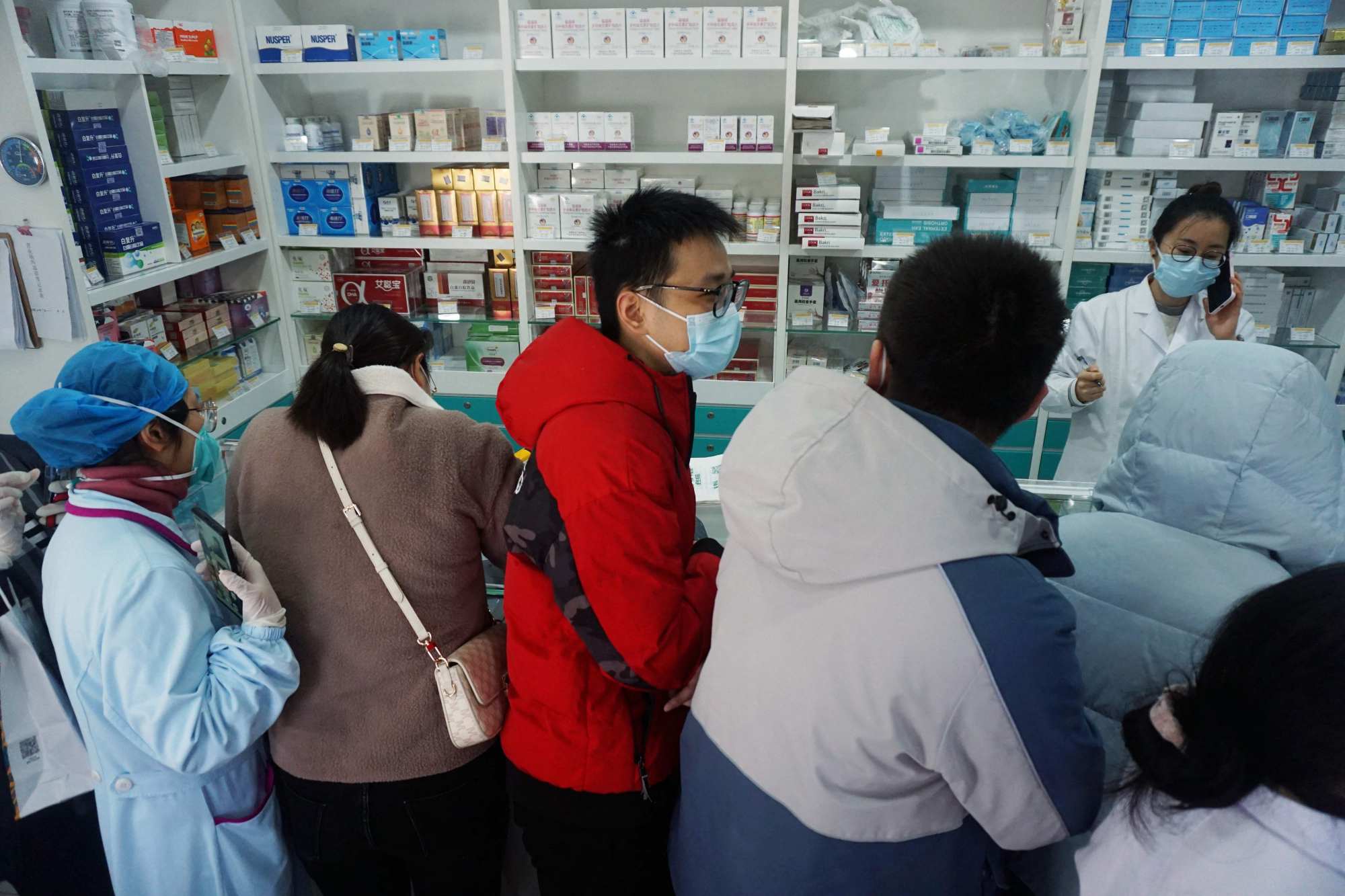
Queues for Covid-19 supplies at a pharmacy in Hangzhou, in China’s eastern Zhejiang province. Photo: AFP
Under China’s drug administration law, medicines marketed abroad but not approved in China are no longer identified as counterfeit, but those involved in their distribution still face administrative penalties for illegal imports without a licence.
Some e-commerce platforms have put the generics up for sale under euphemistic labels to bypass keyword censorship.
Public health experts and doctors in China have warned of the potential risks and urged people not to buy medicines from illegal channels, including unlicensed salespersons on messaging app WeChat.
This comes as surging Covid-19 infections across China spark an acute shortage of pain and fever medication, triggering panic buying at both pharmacies and online healthcare platforms.
“Marketing queries are coming to [Indian] drug makers asking for quotes on ibuprofen and paracetamol,” Sahil Munjal, chairman of the Pharmaceuticals Export Promotion Council of India (Pharmexcil), told Reuters last week.
India would step up production and export of fever medicines to China, he said.
Inside an overcrowded Beijing hospital struggling with Covid surge in China
Capital city Beijing is expected to start distributing Paxlovid and Azvudine to community hospitals, according to state media reports on Monday.
Several such hospitals confirmed to the Post that they had received an official notice for online training on administering the drugs, but stocks had yet to arrive.
Earlier this month, China relaxed the rules to allow retail sales of Paxlovid and also authorised internet healthcare service providers to offer consultations and prescribe drugs, as Covid-19 patients swamped hospitals and clinics.
Paxlovid stocks were sold out within an hour on the mobile app of 111. inc, one such healthcare platform, as people rushed to get a box for 2,980 yuan.
When word got out that a high-end private hospital in Beijing could prescribe Paxlovid, many rushed there to stockpile the drug for the elderly at home, even if the batches were very close to their expiration date.
Among them was Beijing resident May Shen, who said she paid around 3,800 yuan for one pack, including a consultation fee.
She wanted to have the medicine at hand for her father, who has terminal cancer.
“I know the drug needs to be taken under the guidance of doctors, but I am worried that if things get worse we won’t be able to get a hospital bed or see a doctor in time. It’s better to secure the drug now rather than regret it later,” Shen said.
“The drug will expire in February but I am OK with it.”
China gave emergency approval to Paxlovid in February, but it is not clear how much has been imported since then or how widely hospitals are using the drug, which is currently covered by medical insurance.
