WolfPack86
Senior Member
- Joined
- Oct 20, 2015
- Messages
- 10,510
- Likes
- 16,959
Permission was granted after the firm submitted results from pre-clinical studies of the vaccine that demonstrated its safety and immune response, said Bharat Biotech.
The country’s top drug regulator, Central Drugs Standard Control Organisation, has granted approval to Bharat Biotech India to conduct human clinical trials for ‘Covaxin’, making it the first indigenous Covid-19 vaccine candidate to receive this approval, the firm said Monday. These trials are scheduled to start across India in July.
Drug Controller General of India Dr V G Somani approved Bharat Biotech’s application to conduct phase I and II clinical trials for Covaxin, it stated in a release.
The permission was granted after the company submitted results from pre-clinical studies of the vaccine that demonstrated its safety and immune response, said Bharat Biotech in its release. The vaccine was developed in collaboration with Indian Council of Medical Research’s National Institute of Virology.
“The collaboration with ICMR and NIV was instrumental in the development of this vaccine. The proactive support and guidance from CDSCO have enabled approvals to this project,” stated Bharat Biotech chairman and managing director Dr Krishna Ella.
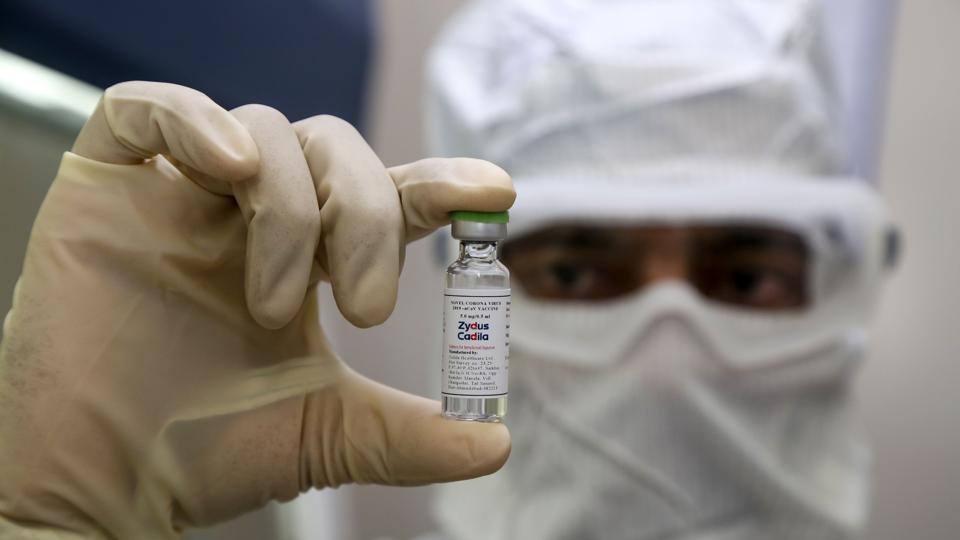
Other Indian firms engaged in the development of Covid-19 vaccines include Zydus Cadila, Serum Institute of India and, since the beginning of this month, Panacea Biotec. While Panacea is still in the pre-clinical stage, it is not clear whether Zydus and Serum have completed their preclinical studies and have also applied to the Central Drugs Standard Control Organisation for approval to conduct human trials yet. Queries sent on Monday night to Zydus and Serum about the status of their vaccine candidates remained unanswered by press time.
Calls to DCGI Somani remained unanswered as well.

 indianexpress.com
indianexpress.com
The country’s top drug regulator, Central Drugs Standard Control Organisation, has granted approval to Bharat Biotech India to conduct human clinical trials for ‘Covaxin’, making it the first indigenous Covid-19 vaccine candidate to receive this approval, the firm said Monday. These trials are scheduled to start across India in July.
Drug Controller General of India Dr V G Somani approved Bharat Biotech’s application to conduct phase I and II clinical trials for Covaxin, it stated in a release.
The permission was granted after the company submitted results from pre-clinical studies of the vaccine that demonstrated its safety and immune response, said Bharat Biotech in its release. The vaccine was developed in collaboration with Indian Council of Medical Research’s National Institute of Virology.
“The collaboration with ICMR and NIV was instrumental in the development of this vaccine. The proactive support and guidance from CDSCO have enabled approvals to this project,” stated Bharat Biotech chairman and managing director Dr Krishna Ella.
Other Indian firms engaged in the development of Covid-19 vaccines include Zydus Cadila, Serum Institute of India and, since the beginning of this month, Panacea Biotec. While Panacea is still in the pre-clinical stage, it is not clear whether Zydus and Serum have completed their preclinical studies and have also applied to the Central Drugs Standard Control Organisation for approval to conduct human trials yet. Queries sent on Monday night to Zydus and Serum about the status of their vaccine candidates remained unanswered by press time.
Calls to DCGI Somani remained unanswered as well.

Bharat Biotech’s Covid vaccine 1st in India to get approval for human trials
Permission was granted after the firm submitted results from pre-clinical studies of the vaccine that demonstrated its safety and immune response, said Bharat Biotech.